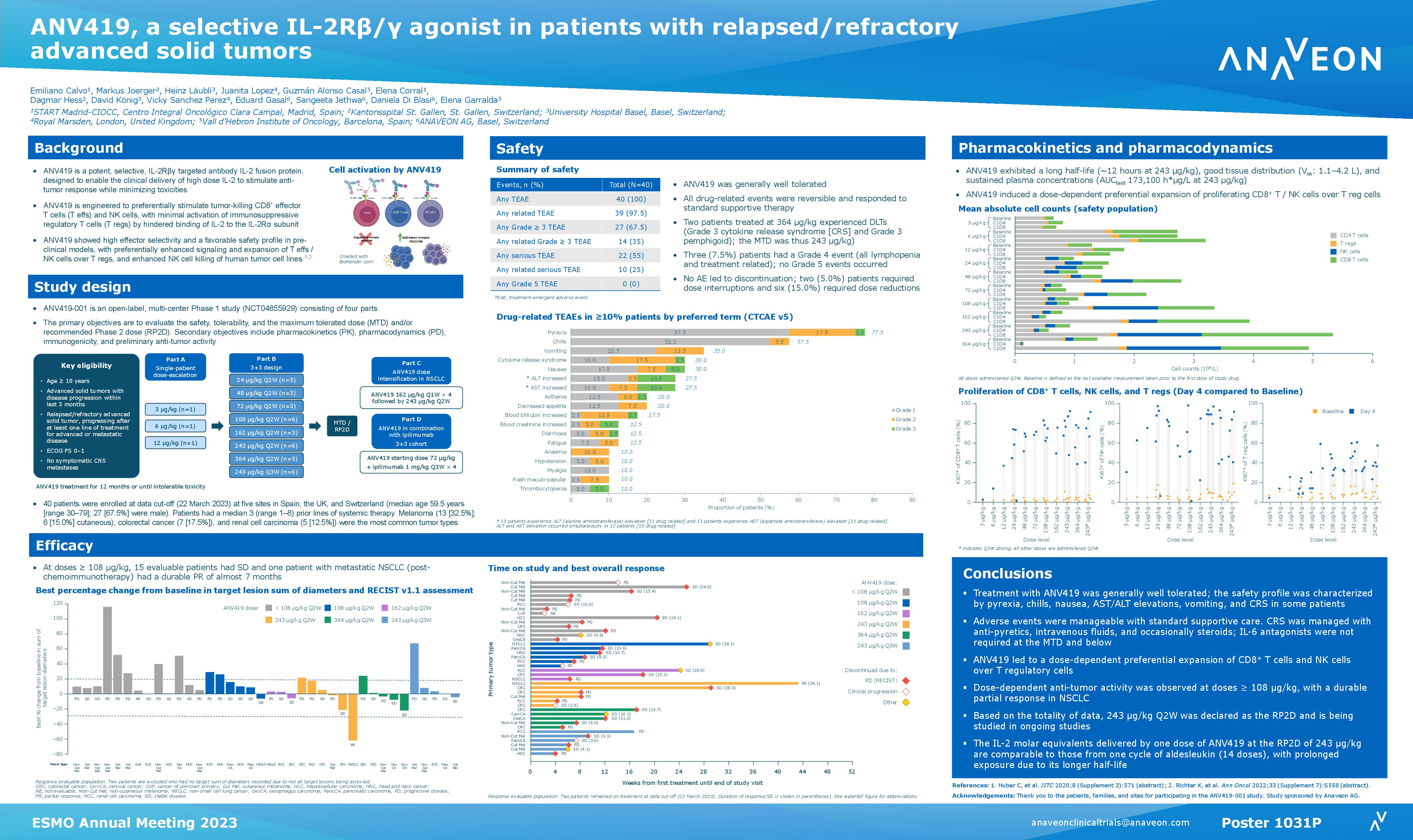
Anaveon announces presentation of ANV600 preclinical data supporting combination therapy with therapeutic PD-1 inhibitors
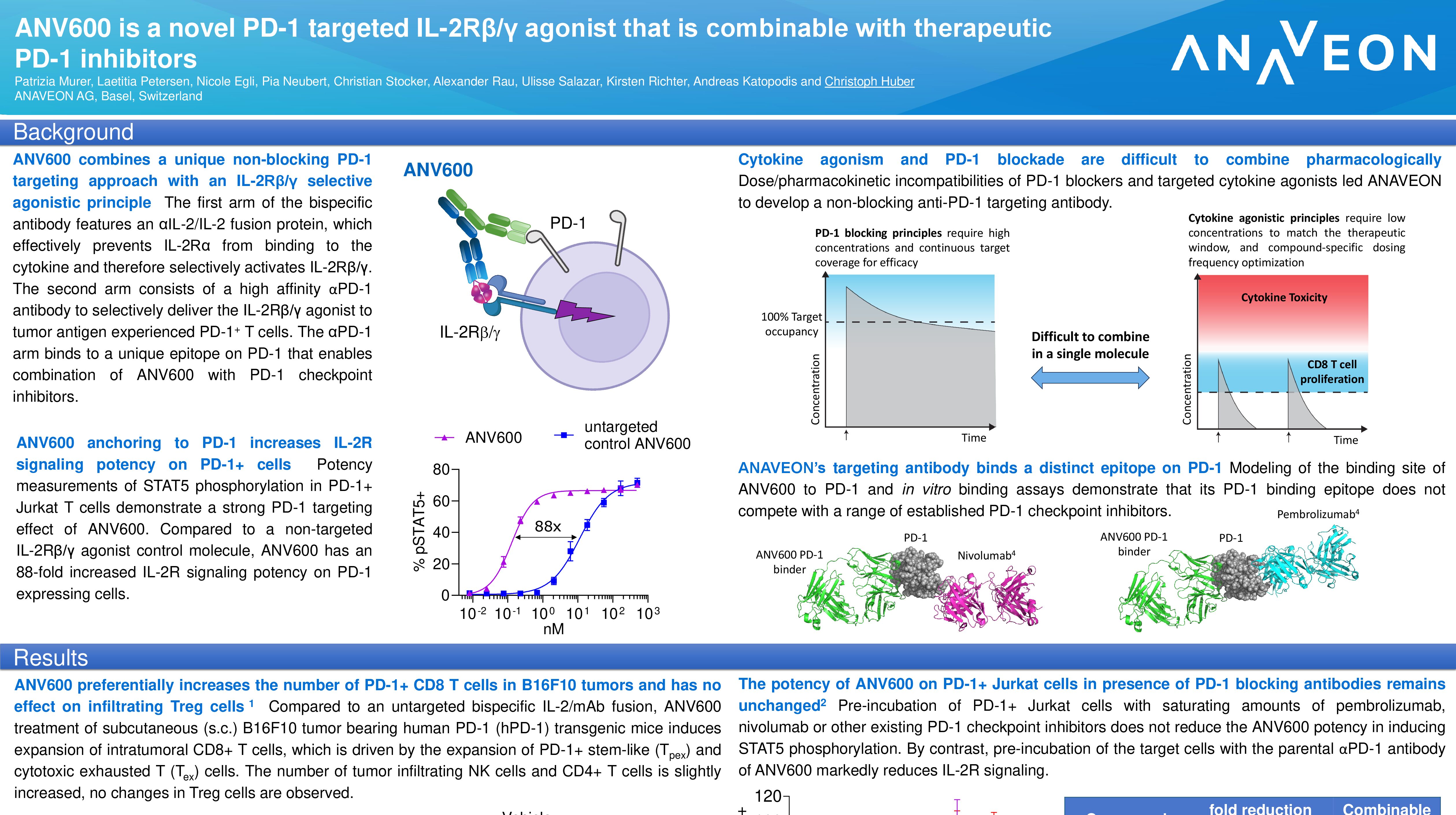
— ANV600 is a potent, cis-signaling, IL-2Rβ/γ agonist which efficiently expands intratumoral stem-like CD8+ T cells —
Basel, November 1, 2023 – Anaveon, a clinical stage, immuno-oncology company, today announced that it will present a poster on ANV600 at the 38th Annual Meeting of the Society for Immunotherapy of Cancer (SITC) being held from Friday, November 3, 2023, to Sunday, November 5, 2023, in San Diego, California.
Targeting of stimulatory cytokines to PD-1 expressing tumor infiltrating lymphocytes (TILs) is a promising approach to reinvigorate antitumor immune responses. Currently, PD-1 checkpoint inhibitors (CPIs) have been established as the standard of care for many cancer indications. ANV600 combines a unique non-blocking PD-1 targeting approach with an IL-2Rβ/γ selective agonistic principle. This is a first example of a PD-1 targeted cytokine agonist which enables therapeutic combination of existing PD-1 inhibitors with novel PD-1 directed stimulatory cytokines.
“ANV600 is a powerful, new, targeted bispecific capable of proliferating tumor specific TILs” said Christoph Huber, Chief Scientific Officer of Anaveon. “We have designed the molecule to be compatible with anti-PD-1 antibodies to allow for optimal therapeutic combination of these agents.”
The abstract is available on the SITC website, and the accompanying poster will be on display on the Anaveon website on November 4, 10:00 am PDT (download poster here).
Details of the poster presentation:
November 4, 9:00 am – 8:30 pm PDT, Poster #820
Title: ANV600 is a novel PD-1 targeted IL-2Rβ/γ agonist that is combinable with therapeutic PD-1 inhibitors
Authors: P. Murer, U. Salazar, N. Egli, L. Petersen, P. Neubert, Ch. Stocker, A. Rau, K. Richter, A. Katopodis and Ch. Huber
Anaveon is developing selective cytokine receptor agonists with the potential to therapeutically enhance a patient’s immune system to respond to tumors. ANV419, currently in Ph II studies in multiple cancer indications, is designed to preferentially signal through the IL-2 beta/gamma receptor resulting in strong proliferation of effector cells in patients. The follow-on compound, ANV600, targets the selective IL-2 receptor moiety to intratumoral effector cells and may have therapeutic benefit in less immunogenic tumors. These novel types of therapeutics, if approved, could potentially have a wide utility in oncology, including in combination with checkpoint inhibitors, cell therapies, vaccines, and radiotherapy.
ENDS
Enquiries:
JW Communications
Julia Wilson
Email: julia.wilson@anaveon.com
Tel: +44 7818 430877
About Anaveon
Anaveon is a clinical stage, biopharmaceutical company, based in Switzerland, that develops biologics to modulate the function of cytokines and provide substantial therapeutic benefit to cancer patients. Our vision is to develop novel immune therapies benefiting patients suffering from a wide variety of diseases with immune pathology. For further information please visit the Company’s website at: www.anaveon.com.